Beta-amyloid plaques and tau tangles are infamous buzzwords in the scientific community, referring to two thought-to-be causative agents of Alzheimer’s disease. However, new research shows that understanding beta-amyloid and tau is only the first step toward tackling the full problem behind Alzheimer’s.
Researchers from the Salk Institute set out to investigate the degradation of apolipoprotein E4 (ApoE4), a protein Alan Saghatelian, PhD, believes to be the “most predictive genetic change for late-onset Alzheimer’s,” a disease which affects more than five million Americans.
Apolipoprotein exists in two other variants aside from ApoE4: ApoE2 and ApoE3. All apolipoproteins are known to carry fat, cholesterol, and vitamins throughout the body, but in terms of Alzheimer’s, each variant seems to play a different part. While ApoE3 seems to have zero impact on disease progression, ApoE2 has been shown to be protective. ApoE4, on the other hand, is the villain of the story.
Past studies have associated a large increase in an individual’s risk for developing late-onset Alzheimer’s with a person containing two copies of the ApoE4 gene. However, scientists know very little about why this relationship exists. “No one has really understood what’s going on at the molecular level,” said Saghatelian. “We’ve found a very different way of thinking about how the proteins in Alzheimer’s disease might be regulated.”
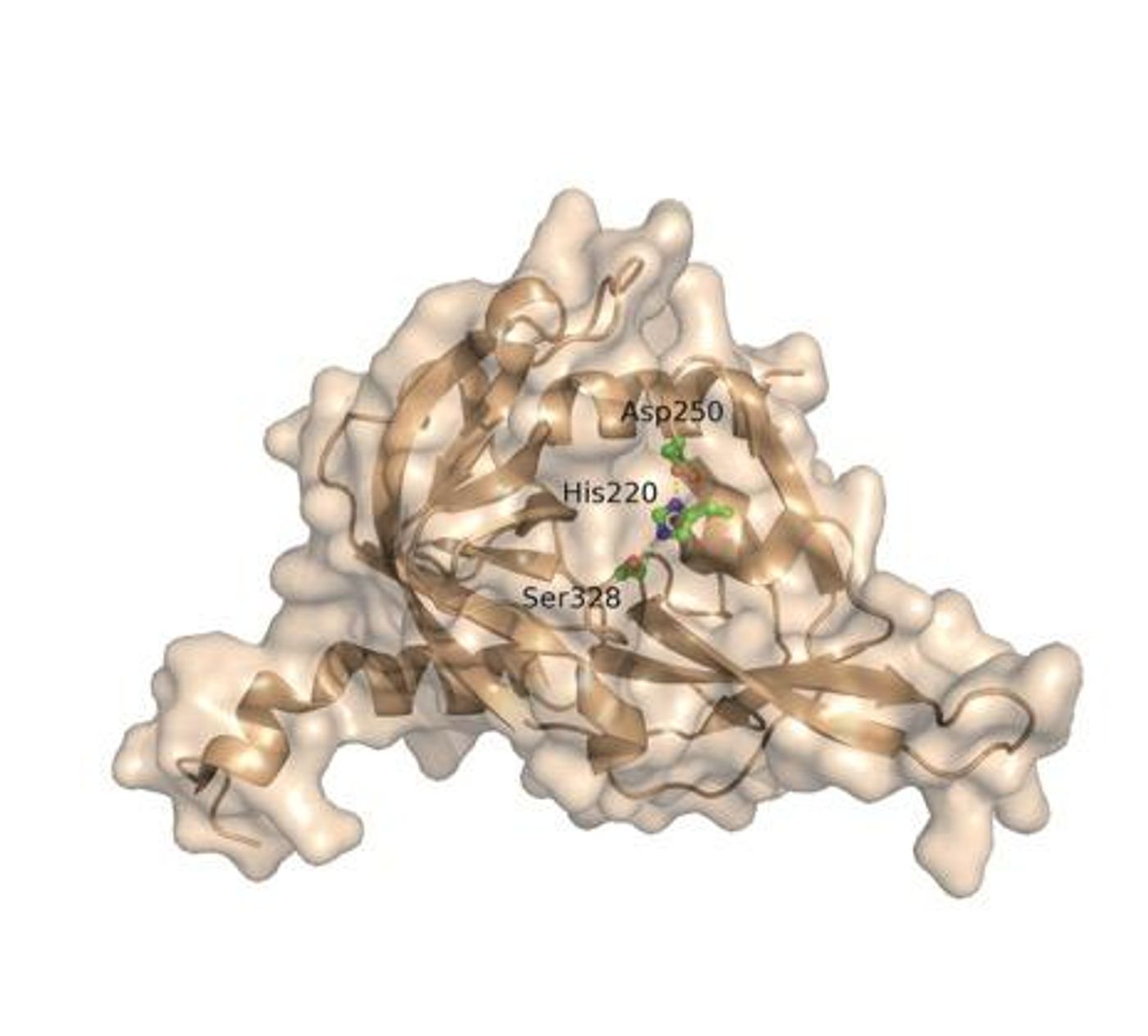
In order to find out whether or not ApoE4 or its breakdown products were toxic in the brain, Saghatelian and his team screened tissues for potential solutions. This led to the investigation of an enzyme called high-temperature requirement serine peptidase A1 (HtrA1), which appeared to be responsible for breaking ApoE4 down into multiple pieces.
Not only was HtrA1 breaking down ApoE4, the enzyme was doing so increasingly more than compared to ApoE3, as the researchers saw in both isolated protein and human cell experiments. By now, they were thinking that people with two copies of the ApoE4 gene were subsequently at an increased risk for late-onset Alzheimer’s because of the enhanced population of ApoE breakdown products in their brain cells.
Meanwhile, after researchers clearly observed the tendency for ApoE4 and HtrA1 to bind, they also realized that their interaction left tau proteins unable to be broken down. With ApoE4 keeping HtrA1 “busy,” the tau protein population grew, creating the tau tangles that are commonly associated with Alzheimer’s.
So maybe it’s not just the tau tangles and beta-amyloid plaques that scientists should be worried about. The new understanding of the relationship between ApoE4 and HtrA1 shows that there may be an indirect cause that should be addressed.
Ultimately, the recent Salk Institute study, published in the
Journal of the American Chemical Society, allows scientists to better understand Alzheimer’s disease while providing helpful information that could one day lead to better ways to develop treatment and prevention methods.
Sources:
Salk Institute,
Alzheimer’s Association