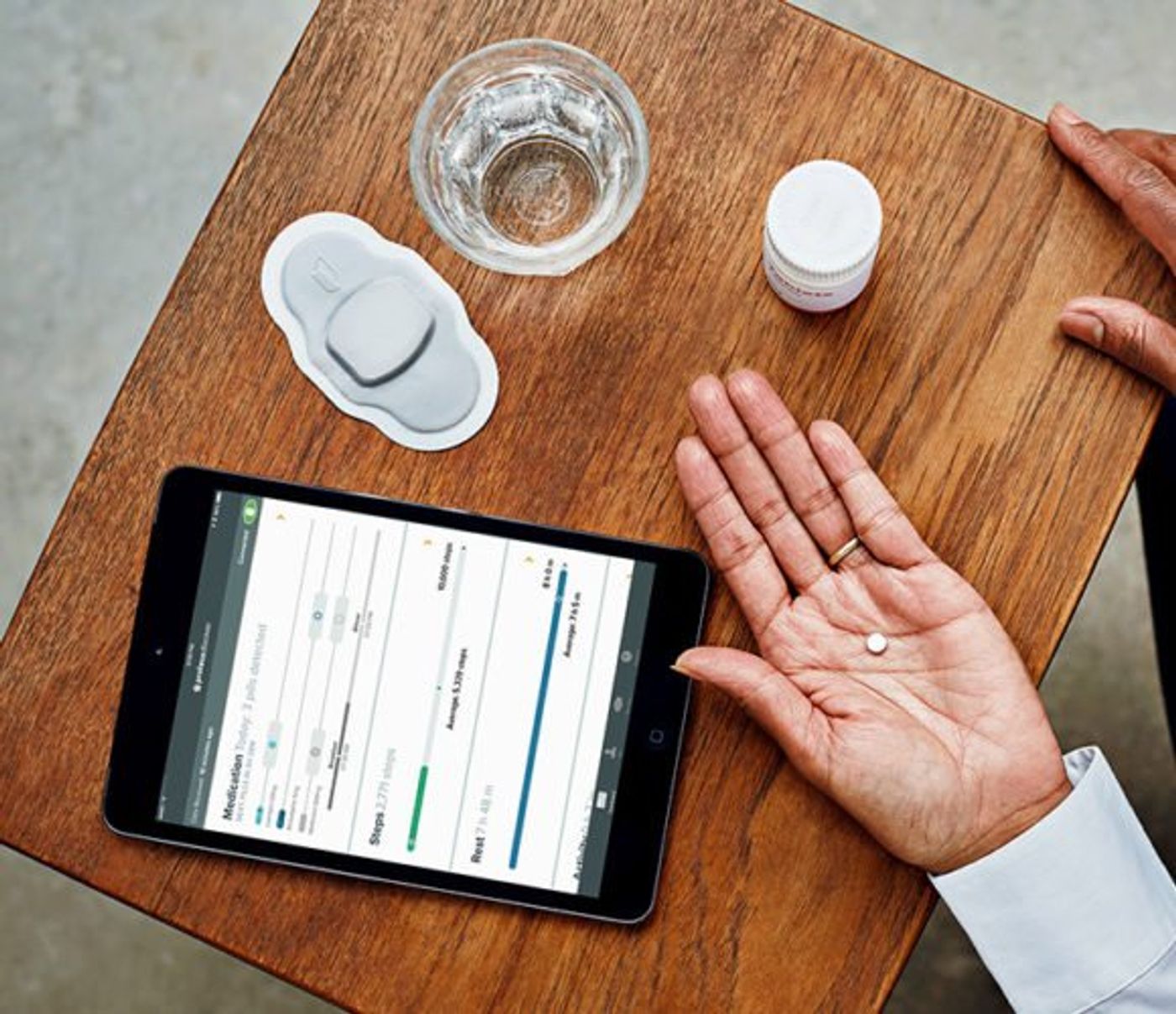
FDA Approves First Digital Drug That Records Being Taken
In Nov. 2017, The Food and Drug Administration (FDA) gave approval to Abilify MyCite, an antipsychotic medication, and the first drug to be approved with a digital ingestion tracking system. This means that the pill contains a small embedded sensor that records when it is taken. The pill sends the date and time it is taken to a patch worn by the patient, which sends it to a smartphone app. This is a major development in digital pill-tracking, an area wherein most previous options have centered on bottles that detect whether they are opened.
"Being able to track ingestion of medications prescribed for mental illness may be useful for some patients," FDA Director of the Division of Psychiatry Products Dr. Mitchell Mathis tells NPR, adding that his organization “is committed to working with companies to understand how technology might benefit patients and prescribers."
Otsuka Pharmaceutical in Japan makes Abilify, which is the brand-name version of the antipsychotic aripiprazole. This medication is often prescribed to people with bipolar disorder, schizophrenia or depression. When one of these individuals takes Abilify MyCite, they ingest a small, grain-sized sensor in the pill. The sensor is made by Proteus Digital Health and becomes active when it hits the stomach fluids.
Dr. Paul Appelbaum, Columbia University’s director of law, ethics and psychiatry in the psychiatry department, tells The New York Times he was surprised to see mental illnesses like schizophrenia be the first conditions to which this high-tech surveillance medicine was applied. "Many of those patients don't take meds because they don't like side effects, or don't think they have an illness, or because they become paranoid about the doctor or the doctor's intentions." Psychiatrist Dr. Peter Kramer warns the Times that this “tattletale” medication “sounds like a potentially coercive tool.”
Patient rights, privacy and confidentiality are prominent themes in any discussion of medical procedure and record, and pill sensors transmitting digital records may bring up unique concerns. "We've seen time and time again that stuff that's being transmitted ends up in the hands of people it shouldn't. There are real concerns about data security," says Dr. Walid Gellad, co-director of the University of Pittsburgh’s Center for Pharmaceutical Policy and Prescribing.
While the drug’s methods and the illnesses’ symptoms may be incongruent in some ways, caregivers might be relieved to have the option of knowing whether a loved one is staying on their prescribed course of treatment, Gellad says. Along with easing some minds, the Times reports that reducing nonadherence (noncompliance to prescriptions) could save a lot of money – it costs billions annually, usually because patients end up needing additional care. Nonadherence can be a dangerous issue for patients with many conditions, including high cholesterol and blood pressure issues.
Abilify can also be given in a monthly injection. According to The Wall Street Journal, the smallest amount of the monthly injectable is $1,478 and a monthly supply of nondigital pills "is at least $891.” The cost of the MyCite version has not been released.
The FDA states Abilify MyCite's hasn't been shown to improve patients' compliance and that it “should not be used to track drug ingestion in 'real-time' or during an emergency because detection may be delayed or may not occur."