Myeloid Cell Differentiation Drives Therapeutic Efficacy
Myeloid-derived suppressor cells (MDSCs) are immune cells that promote chronic inflammatory disease, such as triple negative breast cancer. Consequently, MDSCs are upregulated in triple negative breast cancer and strongly predicts survival outcomes. Therefore, this cell population is critical to study and target to improve patient care.
Myeloid-derived suppressor cells are generated by overexpression of growth factors or proteins, such as granulocyte colony-stimulating factor (G-CSF), granulocyte-macrophage colony-stimulating factor (GM-CSF), and interleukin 6 (IL6). Growth factors alter myelopoiesis, the generation process of myeloid cells, in the bone marrow. The MDSCs, once generated, traffic to the site of infection and promote disease progression by inhibiting various immune cells and releasing cytokines, or proteins, that promote the further generation of MDSCs. Therefore, a positive feedback loop develops, and MDSCs become present in the bone marrow, blood or periphery, and disease site.
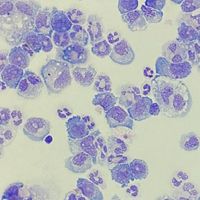
A study published in the Journal of Clinical Investigation (JCI), by Scott I. Abrams and colleagues, discovered that treated MDSCs at the site of origin in the bone marrow could sensitize breast cancer tumors to immunotherapy. The Abrams group treated MDSCs with a dihydroorotate dehydrogenase (DHODH) inhibitor due to its recent application in Acute Myeloid Leukemia (AML), where a DHODH inhibitor known as brequinar (BRQ) differentiates these cells into neutrophils. Interestingly, AML cells and MDSCs share a common progenitor cell, the granulocyte-monocyte progenitor (GMP), which was the rationale for the Abrams group to repurpose BRQ for MDSCs. In comparative models, BRQ was administered daily starting at day 9 with anti-PD-1 monoclonal antibody (mAb) given every other day for three days. As a result, the tumor was significantly reduced in size, and the metastasis to the lung, a common characteristic of breast cancer, was almost non-existent.
To further elucidate the mechanism behind tumor reduction, the Abrams Lab analyzed the MDSCs affected by BRQ. To determine the mechanism of action between the two therapies, in vitro and in vivo models were used. The study shows BRQ matures MDSCs to express a mature morphology and phenotype and downregulation of genes indicative of suppressive MDSCs. MDSCs were also found to have reduced suppressive function demonstrated through a suppression assay where treated- and non-MDSCs were co-cultured with T cells. Moreover, they found similar findings in generated human MDSCs. T cells were also analyzed, and studies showed increased activation of CD8+ T cells. Besides the direct role through anti-PD-1, increased T cell activation was attributed to the differentiation of MDSCs since MDSCS targets essential amino acids necessary for T cell proliferation and activation.
The study demonstrated that an aggressive, highly metastatic cancer model could be treated and significantly reduce tumor size. The Abrams Lab, for the first time, repurposed BRQ for the treatment of MDSCs and has found that it sensitizes tumor growth to immune checkpoint blockade, specifically anti-PD-1. The published findings set a presentence for combination therapy targeting MDSCs as well as T cells which directly target the tumor.