Thermo Fisher Scientific Inc. (NYSE: TMO) is the world leader in serving science, with annual revenue of approximately $40 billion. Our Mission is to enable our customers to make the world healthier, cleaner and safer. Whether our customers are accelerating life sciences research, solving complex analytical challenges, increasing productivity in...
When accuracy matters: MAS Diabetes quality controls
Diabetes the Disease
Diabetes is a chronic health condition that affects the metabolism of individuals. Diabetes is caused by the body’s inability to make insulin or make enough insulin. When there is inadequate insulin produced by the body, blood sugars tend to raise and over period of time with increased sugar in the blood stream, results in various additional health complications including, cardiac conditions, neurological conditions(neuropathy), renal conditions and loss of vision.
According to IDF Diabetes Atlas 20221, one in ten adults living are living with diabetes and half of them go undiagnosed. In 2021, it was estimated that there were over 537 M adults living with diabetes worldwide. The number of individuals with Diabetes is predicted to climb to 643 M by 2030 and 783 M by 2045. Diabetes is also the third health condition outside Cancer and Cardiac that contributes to mortality. In 2021, 6.7 M deaths were reported due to diabetes, 1 in every 5 seconds. Finally, 541 M adults have Impaired Glucose Tolerance (IGT), that places them at high risk of type 2 diabetes.
Importance of Diagnostic Testing and Quality Control for Diabetes testing
Diabetes is a chronic disease that affects the Glucose levels in human plasma. Consequently, the diagnosis of Diabetes are all centered around the detection of blood glucose levels.
The American Diabetes Association has classified individuals in three categories based on the level of Glucose in human plasma.
Normal Levels: Healthy normal individuals typically have Glucose test values in the range of 70 to 100 mg/dl. Blood glucose levels are normally established when the patient or individual is fasting.
Prediabetic: A fasting blood glucose result of 100 – 125 mg/dl (5.6-6.9 mmol/L) indicates that an individual is prediabetic or that they have impaired fasting glucose.
Diabetic: A fasting blood glucose level greater than 126 mg/dl (7 mmol/L) typically indicates the individual is considered to be diabetic.
- The most common diagnostic tests for patients with diabetes are:
- The fasting Blood Sugar Test- Patient blood glucose are evaluated after overnight fasting.
- Oral Glucose Tolerance Test or OGTT is a common test where an individual comes to the lab fasting and is provided a glucose solution (based on body weight) to drink. Period blood samples are drawn and the sugar levels in blood estimated. Concentration of 140 mg/dl at 2 hours is considered to be normal while concentrations of 140 to 199 mg/dl is considered prediabetic and values greater than 200 mg/dl are considered to be diabetes.
- A1C testing – In July 19972 Glycosylation of Hemoglobin was considered to a diagnostic criterion for Diabetes.
The Thermo Scientific Solution
The MAS Diabetes are designed to be used as unbiased independent third-party Quality Control to monitor the performance of A1C assays. The Controls are manufactured in whole blood that closely mimics real patient sample. The Diabetes control DBCL-MP has been tested and are platform agnostic for different automated systems for A1C estimation. The Vista Diabetes control however is specific for use on the Siemens Dimension Vista analyzer platform only.
Routine use of MAS Diabetes control helps provide the lab with information on test variation, reagent lot consistency, operator variation, and can assist in troubleshooting on random or systematic error.
Why Choose the MAS Diabetes Control for Laboratory Testing
Core laboratories are tasked with testing multiple patient specimens during the day and to deliver accurate, consistent, and reproducible test results for better patient management. Thermo Scientific™ MAS™ Quality Controls are independent third-party controls that support you to achieve that goal – while maximizing efficiency every step of the way. They help you streamline routine tasks without sacrificing quality in the process.
- Why use MAS Diabetes third-party control for monitoring the performance of A1C assays.
- MAS Diabetes control offer a Bias-free performance monitoring- The control is not optimized for specific reagents or analyzers.
- MAS Diabetes control matrix is human whole blood based and best mimics the behavior of a patient sample.
- Minimal variations with new lots of manufactured QC.
Clinical laboratories commonly used total allowable error (TAE) as a quality goal. Total allowable error (TAE) specifies the maximum amount of error—both imprecision and bias combined—that is allowed for an assay.
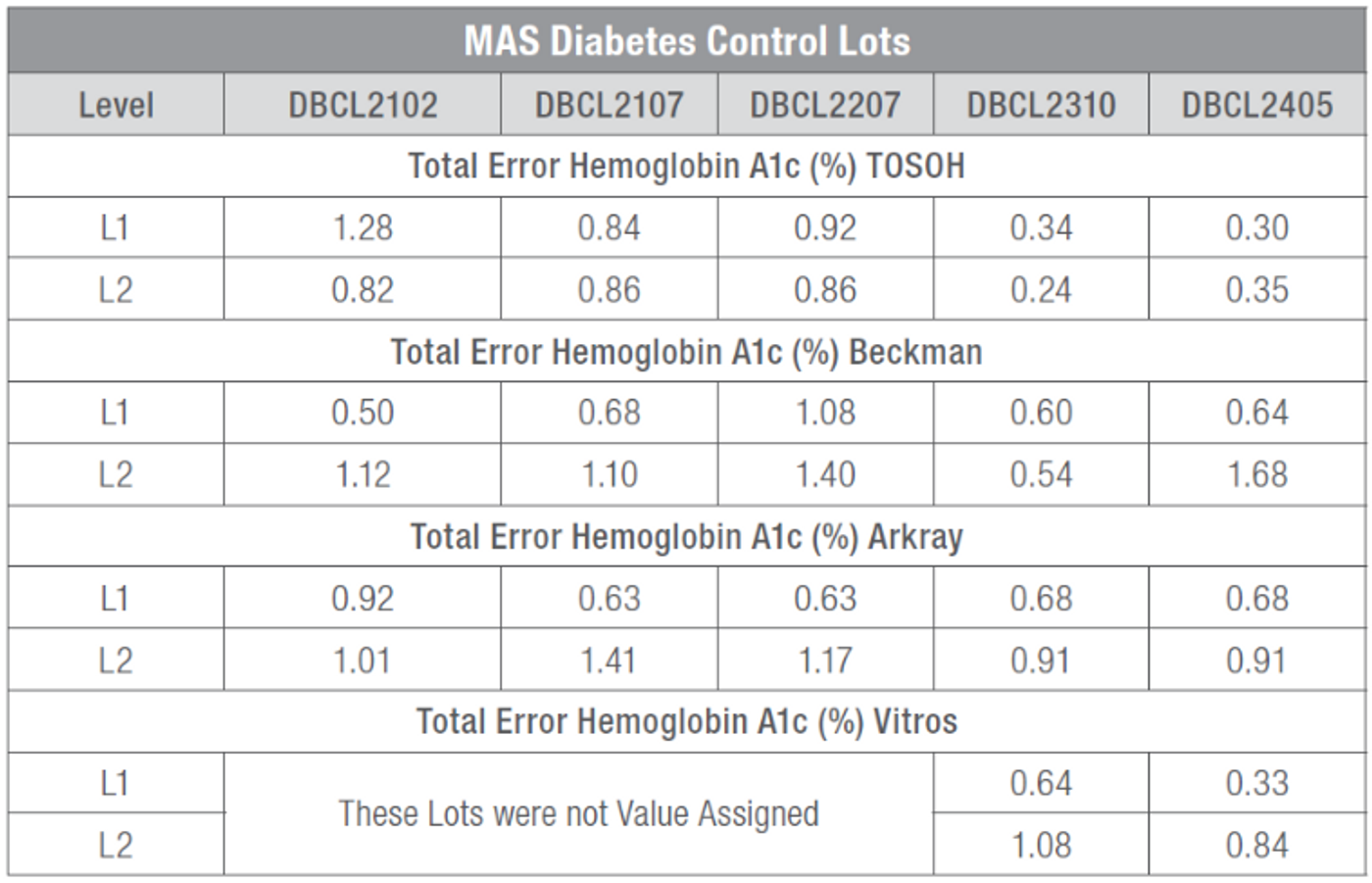
Using the TAE metric an analysis of multiple lots of MAS Diabetes Control released since 2021 was performed. The purpose of this analysis was to calculate the total error observed when testing for the analyte Hemoglobin A1c, in the MAS Diabetes control product. This total error calculation takes into consideration two main contributing factors to variability which are accuracy of measurement (systematic error) and imprecision of measurement (random error). This type of analysis is conducted in a clinical laboratory and on patient samples to determine the accuracy of the test results and significance of the observed change in patient results. The total error can be assessed against limits based on user needs. There are several sources, such as CLIA, NIH and others, available to determine appropriate and acceptable control limits for a particular analyte.
The data used for this assessment consisted of initial instrument-specific Value Assignment (VA) results and compared with customer peer group data imported into LabLink™ software for the same instrument and method. The data points used to compare with VA results were the Lot To Date (LTD) results which were generated over the shelf life of the product as appose to a single time window, such as during a run or during a day. This approach goes beyond the extent of data that would normally be used for such assessment. For example, the standard deviation generated within a run or within a day can be different than the standard deviation generated over the entire shelf life of a product for a particular analyte. A higher level of variability can take place over the shelf life of a product lot than over one run or for one day.
The total error for five (5) lots of MAS Diabetes control was calculated. Results were captured for Tosoh, Beckman, Arkray and Vitros instruments.
All lots evaluated demonstrated acceptable total error results which were well below total allowable error (TAE) limits provided by several sources including National Institute of Health (NIH) and National Glycohemoglobin Standardization Program (NGSP), both having a TAE of + 5 % for Hemoglobin A1c.
MAS Diabetes Controls Product Information
The MAS Diabetes Controls are available in two formats MAS Diabetes control for use on all IVD platforms and the MAS Vista Diabetes Control for use on the Siemens Dimension Platform.
The Diabetes control is a whole blood-based control in liquid, ready to use format. The vials are packaged in a box containing 6 vials of 1 mL each.
Storage Instructions
The MAS Diabetes Controls should be stored at temperatures of -25 to -15 ⁰C. Controls that are unopened can be stored at 2-8⁰ C for 60 days and once a vial is opened it is stable for 30 days at 2-8⁰ C.
Instructions for Use
Thaw control at room temperature (18-25°C) on a rocker or with periodic gentle inversion until liquid and then immediately store at 2-8°C. Thoroughly mix the contents of the vial before each use by gently inverting for several minutes. Open the vial and transfer the required quantity of control into a clean sample cup. Replace cap immediately and store the opened vial at 2-8°C. Assay controls in accordance with the reagent manufacturer’s assay’s and/or instrument’s instructions for unknown specimens.
Limitations
Values for each lot are given in the product insert. Different laboratories, however, may expect slightly different results. It is recommended that each laboratory establish its own expected range. Accurate and reproducible results are dependent upon properly functioning instruments, reagents, and good laboratory technique. This product is intended for use as an assayed control for quantitative assays of listed constituents in human whole blood. This product is not intended for use as a calibrator. For professional use only.
References
1. International Diabetes Foundation. IDF Atlas, 10th edition. Brussels, Belgium: International Diabetes Federation, 2021.
2. Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 1997:20:1183-1197.