A promising treatment for chronic lung infection in cystic fibrosis
Cystic fibrosis (CF) affects over 100,000 people worldwide and is caused by one of several known mutations in the CFTR (cystic fibrosis transmembrane conductance regulator) gene. The CFTR protein is a membrane protein required to transport chloride ions across the cell membrane. Mutations that impair the function of the CFTR protein can lead to an imbalance of ion and water concentration inside and outside the cell. Thus, the mucus secretions from cells containing a faulty CFTR protein become thick and dehydrated.
In normal lung tissue, mucus plays an important role in eliminating pathogens from the airways. The thick and sticky nature of mucus in the lung lining of CF patients provides an ideal environment for the growth of bacteria. Controlling bacterial infection in CF is crucial to slow down lung damage and disease progression. The pathogen Pseudomonas aeruginosa is the most prevalent organism found in the mucus secretions of CF patients and causes chronic lung infection that increases mortality due to CF. The treatment of chronic lung infections in CF is particularly challenging due to antibiotic resistance and frequent bacterial colonization of the mucus lining in the lungs.
Image via Wikimedia Commons Creative Commons Attribution-Share Alike 3.0 Unported license.
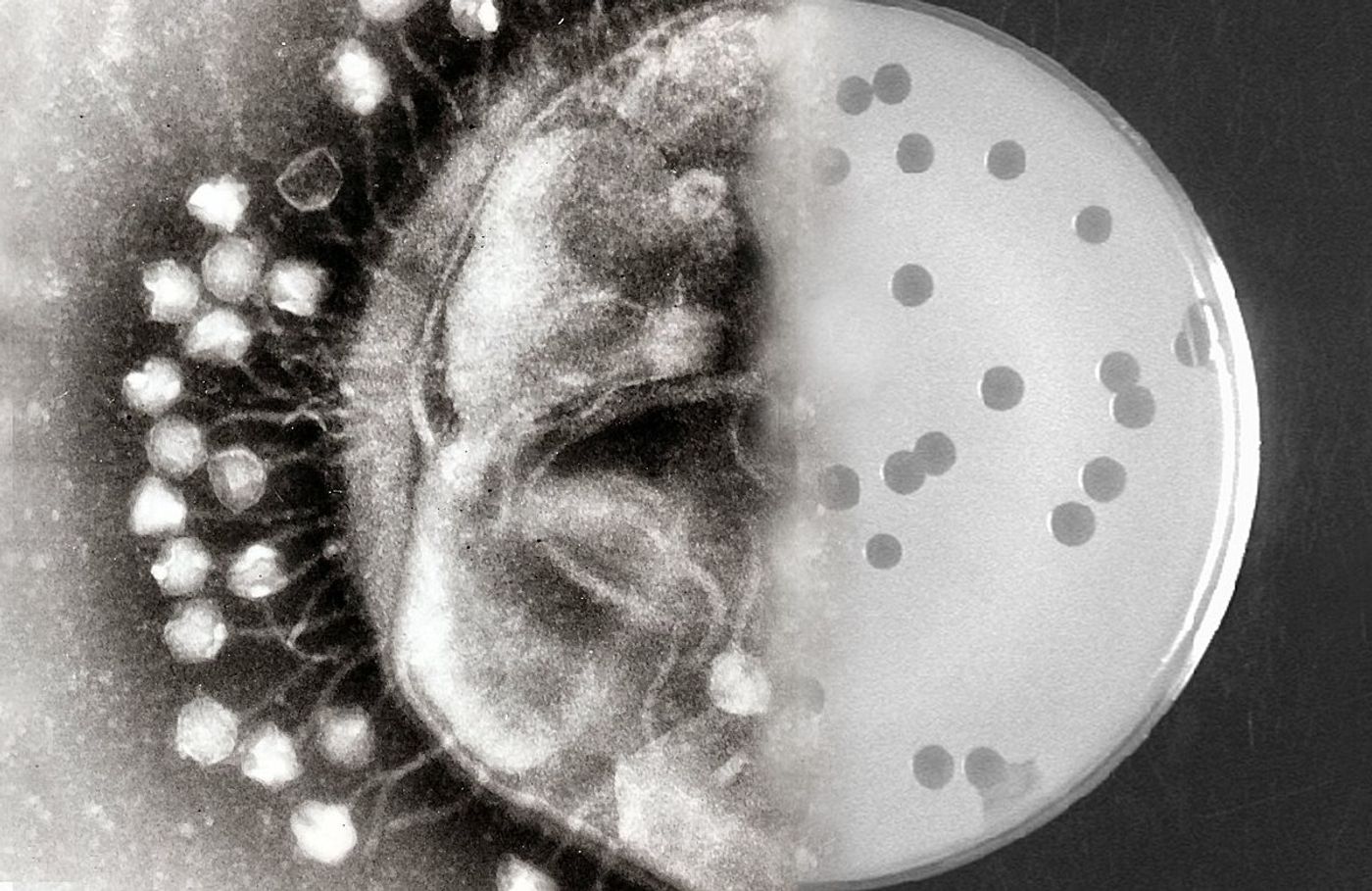
A new, experimental treatment option is being investigated for CF patients that carry P. aeruginosa in their mucus lining. A clinical trial supported by the National Institute of Allergy and Infectious Diseases (NIAID) is studying the effectiveness of phage therapy. Phage therapy utilizes viruses specific to bacteria (called phages) to treat bacterial infections. Phages (also known as bacteriophages) are viruses that infect bacteria. Phages are diverse and abundant, but the replication of a bacteriophage is highly specific to its host bacterium. Phage therapy, therefore, can utilize this specificity to naturally infect and kill bacterial cells within a site of infection. The clinical trial, led by the Antibacterial Resistance Leadership Group (ARLG) at the NIH, will test a cocktail of four different phages that naturally infect P. aeruginosa. The phages will only target Pseudomonas, and human lung cells will remain unaffected.
The clinical trial will take place in two phases: Phase Ib of the study will check for the safety of the phage infusion in a small group of CF patients who carry P. aeruginosa in their lungs. Once the safety data of the phage cocktail is verified, Phase II of the study will recruit a larger group of patients to study the efficacy of the phage infusion as therapeutic. A control group of subjects will receive placebo treatment in Phase II. Due to their self-replicating nature, the long-term effects of phage are still under consideration. NIAID Director Anthony Fauci has said that “although research on bacteriophage therapy may still be in its infancy in the United States, we hope that this study, and others like it, could open the doors to a new type of therapy for difficult-to-treat bacterial infections.” The use of phage therapy in CF is promising, and this clinical trial could serve as a paradigm for treatment of other bacterial infections using phage therapy.
Sources: EurekaAlert!, Antibacterial Resistance Leadership Group (ARLG)