Best practices for lateral flow assay development
In our best practices blog, we explore how lateral flow assay design affects test performance. Read on to gain a deeper understanding of the development process, enabling you to streamline the development of your diagnostic test.
Lateral flow assays (LFAs) provide us with a rapid and convenient method of detecting or measuring the concentration of an analyte from a sample. LFAs are widely used for point-of-care diagnostics. Most LFAs are immunoassays, which rely on the high affinity of antibodies for an analyte epitope to produce a signal.
When designing your own lateral flow assay, there are several variables to consider, with choice of detection reagents, assay materials, and manufacturing methods all influencing the accuracy and reliability of your test.
In this best practices blog, we highlight ten important considerations when developing your LFA, which we hope will give you a deeper insight into lateral flow assays to streamline your development process.
1. Know your target molecule
The biology of your target molecule will be the key factor in developing the biology of your test. Is it a large or small molecule? What antigen or other ligand will bind specifically to your target, but not to other molecules? The kinetics of the binding reactions between your analyte and the antibodies used in the test will determine the optimal flow time of your assay, which will in turn influence your choice of nitrocellulose (NC) membrane. A keen understanding of the biology of your target molecule will shape every choice in the design of your LFA
2. Know your sample type
The biological source of your sample will influence the design of your test, and help you determine which materials you should use for in your assay.
The type and volume of liquid sample (e.g., blood, urine, or saliva) are two parameters that will help you choose which sample and absorption pads are most appropriate for your test.
Start developing your lateral flow immunoassay with our diagnostic services
3. Pick your test type
There are two approaches to lateral flow assay tests, sandwich and competitive, as shown in Figure 1.
Fig 1. Comparison of sandwich and competitive lateral flow assay design.
The size of your target molecule will be a key factor in determining the type of LFA you choose.
- Sandwich assays rely on the binding of two separate antibodies to different regions of the same analyte molecule, making them well suited for detecting high molecular weight analytes.
- Competitive assays can detect smaller molecules such as mycotoxins or cortisol, as well as larger analytes such as insulin.
4. Materials matter!
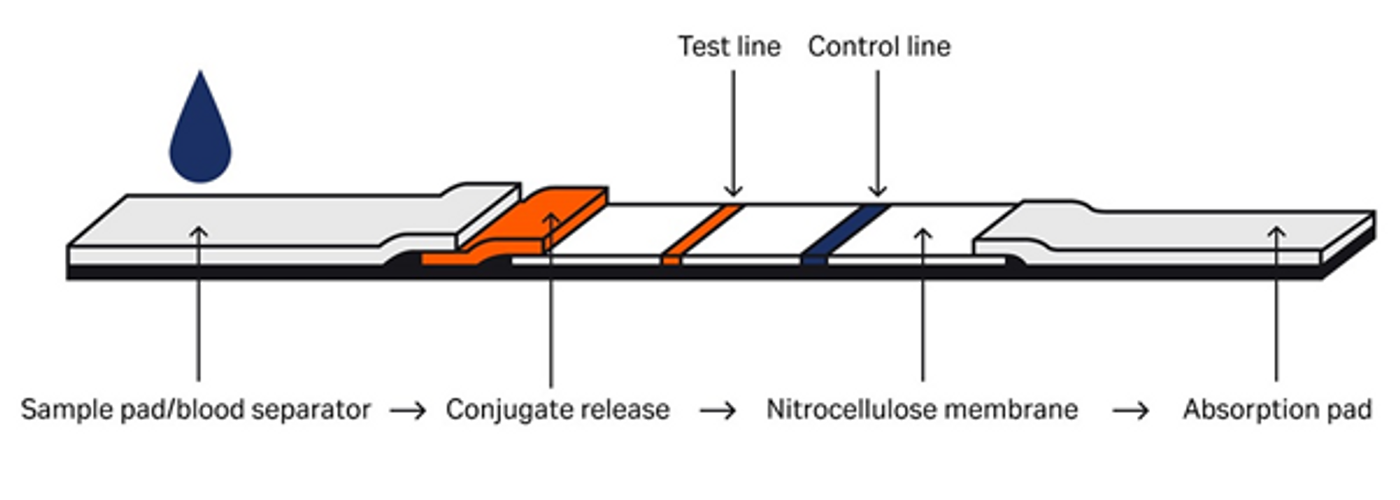
Choosing the right sample pad, conjugate release pad, and nitrocellulose membrane is critical to developing sensitive and reliable lateral flow assays.
Sample pads are commonly made from either cotton or bound glass fiber. Cotton has a lower wicking rate and is suitable for low sample volumes. Bound glass is appropriate for separation of red blood cells (RBCs) from plasma. Using glass fibers with a higher diameter can also prevent hemolysis, the bursting of RBCs that turns plasma red and prevents accurate assay readouts.
It’s important to determine that the conjugate release pad you choose will store the detection reagents without damage or aggregation over the shelf life of the test strip. Choosing hydrophilic materials with an open structure can support efficient liquid flow through your immunoassay.
Discover our guide to building a smarter diagnostic assay
Fig 2. Lateral flow assay components
5. Test membrane flow rates
Nitrocellulose (NC) membranes are one of the most important components of a lateral flow assay; their interactions with your sample will directly affect test performance.
To help ensure a good balance between assay speed and sensitivity, try testing the flow rate of your sample with a range of different NC membranes. The membrane you choose should allow adequate time for interaction of analyte and reagents at the test and control lines, but also provide results within your chosen timeframe.
6. Test, and test again!
NC membranes contain surfactants that could denature analyte proteins and affect antibody-antigen binding reactions. So, testing your detection and capture reagents on a range of membranes with different surfactants before you decide which is the best membrane for your test is a good practice.
Find out more about membrane selection for lateral flow immunoassays
7. Monoclonal or polyclonal antibodies?
The detection and capture antibodies in your lateral flow assay can be polyclonal or monoclonal.
Monoclonal antibodies are generally preferred as capture antibodies, as they have minimal batch-to-batch variability and are less prone to problems in supply than polyclonal antibodies.
Monoclonal antibodies are also the preferred detection reagents, as their recognition of a single epitope provides a consistent binding reaction with minimal cross reactivity. Polyclonal antibodies can be unsuitable for detection as their recognition of multiple epitopes might lead to antigen cross-linking that can prevent analyte from entering the NC membrane.
8. Maximize your assay sensitivity
After choosing antibody binding regions on your analyte, the next step involves screening the set of potential antibody clones and measuring the amount of antibody-antigen complex formed after incubation. But what’s the best way to do this screening step?
- ELISAs are a common way of screening antibodies. However, the long duration of this assay means that it cannot identify antibodies that bind quickly to the target antigen, which is an essential requirement for high-sensitivity assays.
- Using optical biosensors, such as surface plasmon resonance (SPR) or bio-layer interferometry (BLI), can give you a more accurate measure of antibody binding kinetics and help you maximize the sensitivity of your lateral flow assay.
Read our knowledge article, The path to high-quality immunoassay
9. Understand your detection method
There are many different labels that can be conjugated to detection antibodies. Which do you choose?
While fluorescent molecules and enzymes can work well as labels, some of the most widely used are those that produce a direct, visible signal, such as gold nanoparticles and latex beads. As well as having good optical characteristics, gold nanoparticles have good antibody compatibility and long-term stability, important benefits when your tests might be stored for extended periods.
Your choice of label will determine your choice of reader — and vice versa. For visible labels, electronic optical readers can measure analyte concentration, with detectors available in benchtop and handheld formats. Recent developments have also led to systems that provide analysis software in convenient smartphone apps for visible detectors such as gold nanoparticles.
Whichever label you decide on, by testing your assay-reader combination with known concentrations of analyte you can measure the lower and upper limits of quantification and obtain the reader’s measuring interval. Take these limits into account to minimize the risk of under- or overestimating the sensitivity of your lateral flow assay.
10. A final word on testing
As you’ve probably figured out by now, iterative testing is part of the development process. This can be time consuming, but an experienced LFA services provider can help shorten the development cycle and support you with efficient methodologies, infrastructure, and knowledge gained from experience.
What next?
Lateral flow assays are often a first point of call when developing rapid diagnostic tests that require little or no user training. Their ease of use and reliability has made LFAs well-established as home-based pregnancy tests, and recently they have proved crucial in the detection of SARS-CoV-2 and controlling the spread of COVID-19.
Understanding how your sample liquid and analyte molecule will interact with the LFA components can save you time and effort during test development.
For more guidance on generating reliable lateral flow assays, read our blog on custom lateral flow assay development.
To discuss any challenges you might be facing with lateral flow assay development, our diagnostics specialists are ready to help you.
Contact a specialist, or request a sample
Related content:
- Cytiva page: Lateral flow immunoassay development services
- Cytiva guide: Helping you build a smarter diagnostic assay
- Cytiva blog: Membrane selection for lateral flow immunoassays
- Cytiva blog: Lateral flow assays and precision diagnostics
- Cytiva how-to-guide: Multiplexing your lateral flow assay
- Cytiva page: Custom lateral flow assay projects
- Cytiva page: Detection of coronavirus (COVID-19)