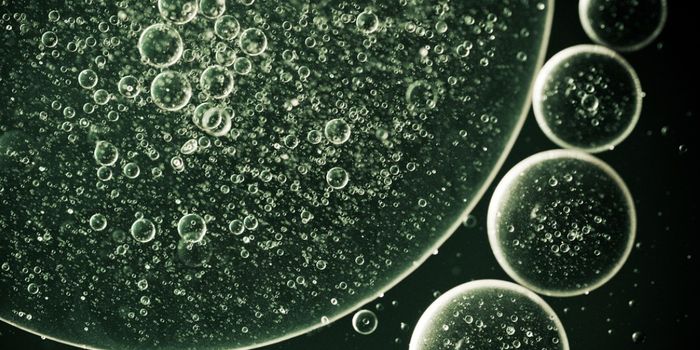
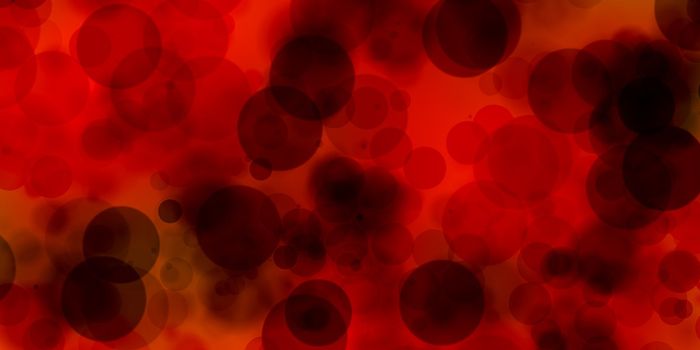
The liquid-liquid phase separation, observed when a crystallization reagent is added to a protein resulting in liquid drops, is also known as “phase separation”. These phase separation droplets are known to contain a high concentration of protein. When these droplets are centrifuged, or simply over time, the drops may separate completely from the rest of the solution, forming two separate liquid phases. The concentration in each of the two phases will be different. These quantity and size of these droplets can vary depending on the conditions of the environment and this separation may exist for days, weeks or months. Phase separation is known to influence the function and activities of proteins, specifically its role in causing disease.
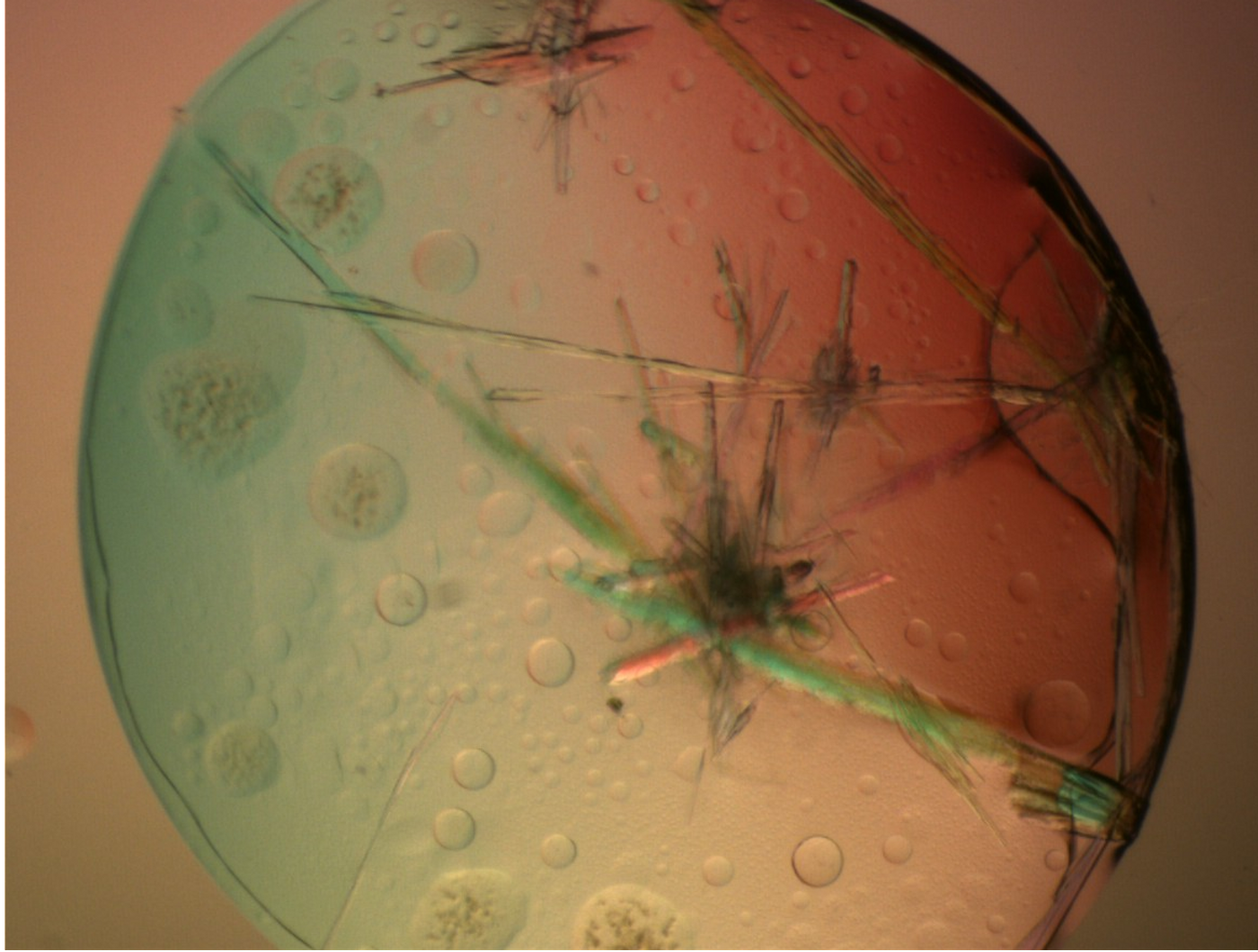
Phase separation is known to influence the function and activities of proteins, specifically its role in causing disease.
Image Credit: Xtal-Concepts
According to a review published this month in
Nature Reviews, phase transition is important for protein functions during cell replication specifically microtubule polymerization and spindle fiber assembly. Proteins which contain unstructured regions can undergo phase separation leading to droplet formation. For example the authors discuss the vertebrate protein BuGZ, which is a component of the spindle matrix, formed droplets due to the fact that the genomic sequence of this protein has low amino acid complexity and is predicted to be unstructured. Microtubule binding by BuGZ was shown to facilitate phase transition at low protein concentrations. It was discovered that microtubule binding promotes BuGZ droplet formation which allows local concentration of BuGZ molecules to further facilitate spindle assembly during cell replication.
Another study highlighted in this review described the role of protein phase separation in the assembly of RNA-protein granules (RNP). RNPs are involved in many important non-membrane-bound cell organelles such as the nucleolus. Many RNPs contain unstructured, low complexity regions they are able to undergo efficient droplet formation. Similarly to microtubule binding in BuGZ, RNA binging in RNP proteins allows efficient interaction between proteins in order to drive droplet formation. Researchers conclude that droplet formation by RNP proteins increases their potency to undergo fibrillization which has previously been associated with severe human degenerative diseases.
Through the accumulation of this research we have observed how proteins involved in different important cellular functions can undergo phase separation to form liquid droplets. Scientists are interested in learning more about the cellular protein phase separation process and how it can be altered in order to prevent disease.
Sources:
Nature Reviews;
Hampton Research